 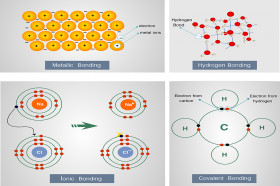
Sodium now has 8 electrons in its outer shell and is stable. When these two elements react, sodium gives the one electron in its outer shell to chlorine. 
Chlorine, on the other hand, has an atomic number of 17 and has 7 electrons in its outer shell. Sodium has an atomic number of 11, hence, sodium has one electron in its outer electron shell. To explain how ionic bonds form we will use common table salt, NaCl, as an example. Hence, H 2 (same element) is a molecule, and H 2O (different elements) is both a molecule and a compound. 
Molecules composed of more than one type of element can also be called compounds. Molecules are two or more atoms held together by the sharing of electrons (described below). In some cases this involves the formation of molecules. The processes that result in the filling of the outer electron shells result in the formation of chemical bonds. The sections below describe some of the important processes by which atoms become stable. The atoms of all of the other elements have vacancies in their outer electron shells and will react with other atoms to fill their outer shells. Chemists often refer to the “rule of 8” which states that if there are 8 electrons in the outer electron shell, the element is stable. These elements are all gasses and they are all stable, meaning that they do not react with other elements. helium, neon, argon, etc) have 8 electrons in their outer shells (with the exception of helium), hence their outer electron shells are full. The branch of chemistry dealing with carbon compounds is called organic chemistry.Close examination of the periodic table will show that the atoms of all of the elements in the last column of the table (i.e. Liacos explains how a molecule's structural formula is constructed based on its chemical formula. Liacos explains how to measure the mass of an amount of gas. Compounds made only of carbon and hydrogen atoms are called hydrocarbons. Chemical Formulas (04:20)Īlexander Butlerov suggested, in the late 1850s, that atoms can form double bonds. Scientist Edward Frankland, in 1852, first suggested that all atoms form a specific number of bonds. Liacos explains how the structural diagram was used prior to the discovery of electrons. All group elements in the periodic table have the same number of valence electrons. Liacos builds a structural diagram of a nitrogen atom. In 1919, Irving Langmuir introduced the word covalence. Eventually, Niels Bohr developed his theory of electron shells. The idea of covalent bonds was introduced by a U.S. Gaseous nitrogen is an example of a triple bond. Liacos demonstrates how atoms can share more than one electron, using gaseous oxygen. When metals chemically react with non-metal atoms, they typically form ionic bonds. Ionic Bonds and Alloys (02:12)Ĭovalent bonds usually occur between the atoms of non-metals, including metalloids. He explains the importance of depicting the shared elections, and what an inaccurate electron dot diagram looks like. Liacos draws an electron dot diagram for ammonia, NH3. Covalent bonding is often described as a sharing of electrons. Liacos explains how the valence shell plays an important role in covalent bonding, using an electron dot diagram. Whenever atoms bond covalently with other atoms, they end up with eight electrons in their outer shells. Liacos examines how covalent bonding occurs in water molecules. A molecule is two or more atoms held together by covalent bonds. Liacos uses a chemical reaction between hydrochloric acid and magnesium to demonstrate covalent bonding. Atoms join into compounds in two ways, a covalent bond or an ionic bond. 
Narrator, Spiro Liacos, explains how the discovery of electron shells led to an understanding of atom bonding. In chemistry, a compound is made of two or more different types of atoms that have chemically joined together. Compounds and Molecules (02:11) FREE PREVIEW 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
